>We support
digital transformation
on the SAP Platform
Offer
SAP Projects & Services
Digital Transformation
KBJ Proprietary Solutions
Our Experts
Get the facts
We have completed over 600 SAP projects over 17 years.
The majority of the team are experts with many years of experience in the SAP market.
More than 90 international SAP certificates confirm our commitment to continuous investment in team knowledge and competence development.
We provide solutions that are an everyday work tool for hundreds of thousands of people.
KBJ experts implement over 30% of all SAP trainings in Poland!
The first SAP partner listed on the stock exchange
Your intelligent enterprise
Find out about the - above the market average - profit plan
Join intelligently managed companies
Be ready to scale your business fast and easily
Perform a smooth, hassle-free digital transformation
Our latest product
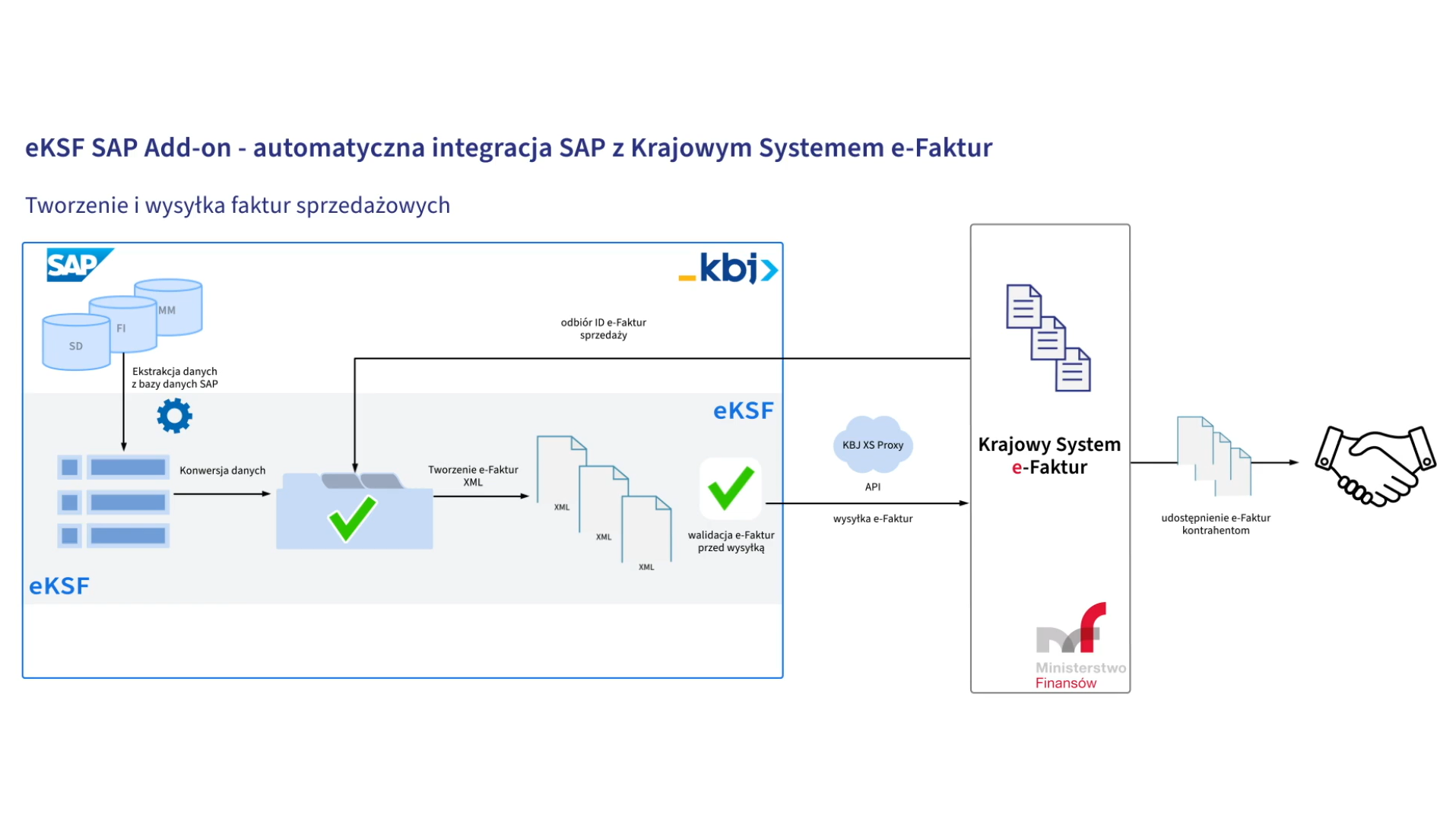
e-KSF
e-KSF is a friendly manager with a number of functions allowing the user to efficiently manage the invoice recording process in the enterprise.
e-KSF enables easy integration of SAP with the National System of e-Invoices (pl.: KSeF) and automatic handling of the process of sending and receiving structured invoices.
From the second quarter of 2023 structured invoice will become the only form of invoicing, and the use of the National e-Invoices System in Poland will become mandatory.
Find out why the e-KSF solution is right for your company!